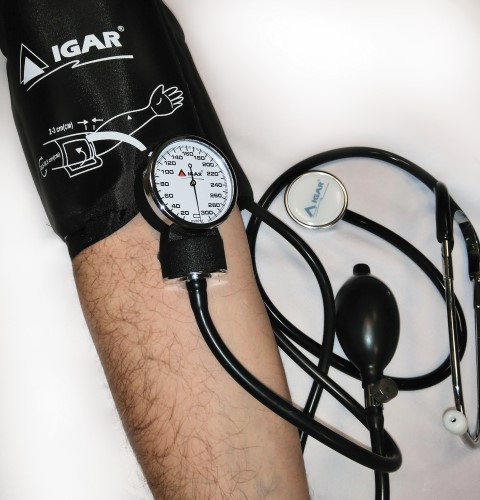
Aneroid sphygmomanometer with stethoscope trade mark IGAR
Aneroid sphygmomanometer with stethoscope trade mark IGAR is a high-quality and accurate sphygmomanometer with improved mechanism.
There is a complete set in the kit and the device is ready for use. Designed to measure blood pressure at home and in medical conditions.
There is a complete set in the box, the device is ready for use.
Aneroid sphygmomanometer is a necessary device for every family!
Аdvantages:
- High quality аneroid sphygmomanometer in steel body frame
- Metal reliable valve with smooth descent
- Balloon cuffs and bulb are made from high quality latex with a seamless technology.
- Simple, accurate and convenient to use.
- The optimal price/quality ratio
Specifications:
- pressure measurement range, mmHg: 20-300;
- minimal measuring step: 2 mm. Hg
- pressure measurement accuracy: + -3 mm Hg
- weight: 385 g
The kit also includes a purse-case with a clasp, made of high-quality nylon, for storing the sphygmomanometer and for its safe transportation.
High accuracy sphygmomanometer is in a steel casing. The metal reliable valve has a smooth descent. All tubes of the device are from latex of necessary convenient length.
Balloon cuffs and blower pears are made from high quality latex using a seamless technology.
Cuff size 28,5-38,5 cm. This cuff is suitable for most adults. The cuff has a retaining ring, making it even more convenient to use.
Metal stethoscope with high-quality sensitive membrane and soft ear olives.
Packing/Morion Code:
Instrument grade:
cuff with internal latex camera, medical manometer, rubber bulb (pear) with a valve, bag-case, metal stethoscope, instruction manual, key.
The manufacturer reserves the right to change the technical characteristics of the device.
Morion Code: 535629
Warranty: 12 months
Storage conditions:
- Storage temperature: from – 20 ° С to + 70 ° С
- Storage humidity: 15-85% non-condensing
- Operation temperature: from + 10 ° С to + 40 ° С
- Operating Humidity: 15-85% non-condensing
Important!
– Do not drop the device.
– Do not expose the device to too high / low temperatures, humidity or direct sunlight.
– Do not touch the fabric from which the cuff is made with sharp tools.
– Store the cuff with completely released air from it.
– Do not disassemble the pressure gauge.
– Store the device in a bag to keep all the parts clean.
Manufacturer: “IGAR Ltd”,Ukraine
Certification:
The medical device is declared and placed on the market of Ukraine according to the requirements of Technical regulations and Conformity Assessment of medical devices, the product has a complete package of documents and packing labelling in Ukrainian language according to the national standards and Technical regulations.
Quality management system of the Manufacturer meets the requirements of DSTU EN ISO 13485.
Complies with the requirements of the Technical regulations regarding medical devices and the requirements of the Technical regulations of legally regulated measuring equipment.
IGAR Declaration No. 31-08 / 2018 dated November 08, 2018 (revision 2) on compliance with the requirements of the Technical regulations regarding medical devices and declaration of November 8, 2018 compliance with the requirements of the Technical regulations on legally regulated measuring equipment, approved by a resolution of the Cabinet of Ministers From January 13, 2016, which is a single declaration of conformity.
The declaration was made under the full responsibility of Igar Ltd., which is responsible for the placement of the medical device on the market.
| Размеры | Норма упаковки, шт/пар | Код Морион | |
|---|---|---|---|
| Групповая | Транспортная | ||
|
XS
(5-6) |
100/50 | 100/50 | 478023 |
|
S
(6-7) |
100/50 | 100/50 | 179220 |
|
M
(7-8) |
100/50 | 100/50 | 179221 |
|
L
(8-9) |
100/50 | 100/50 | 179222 |