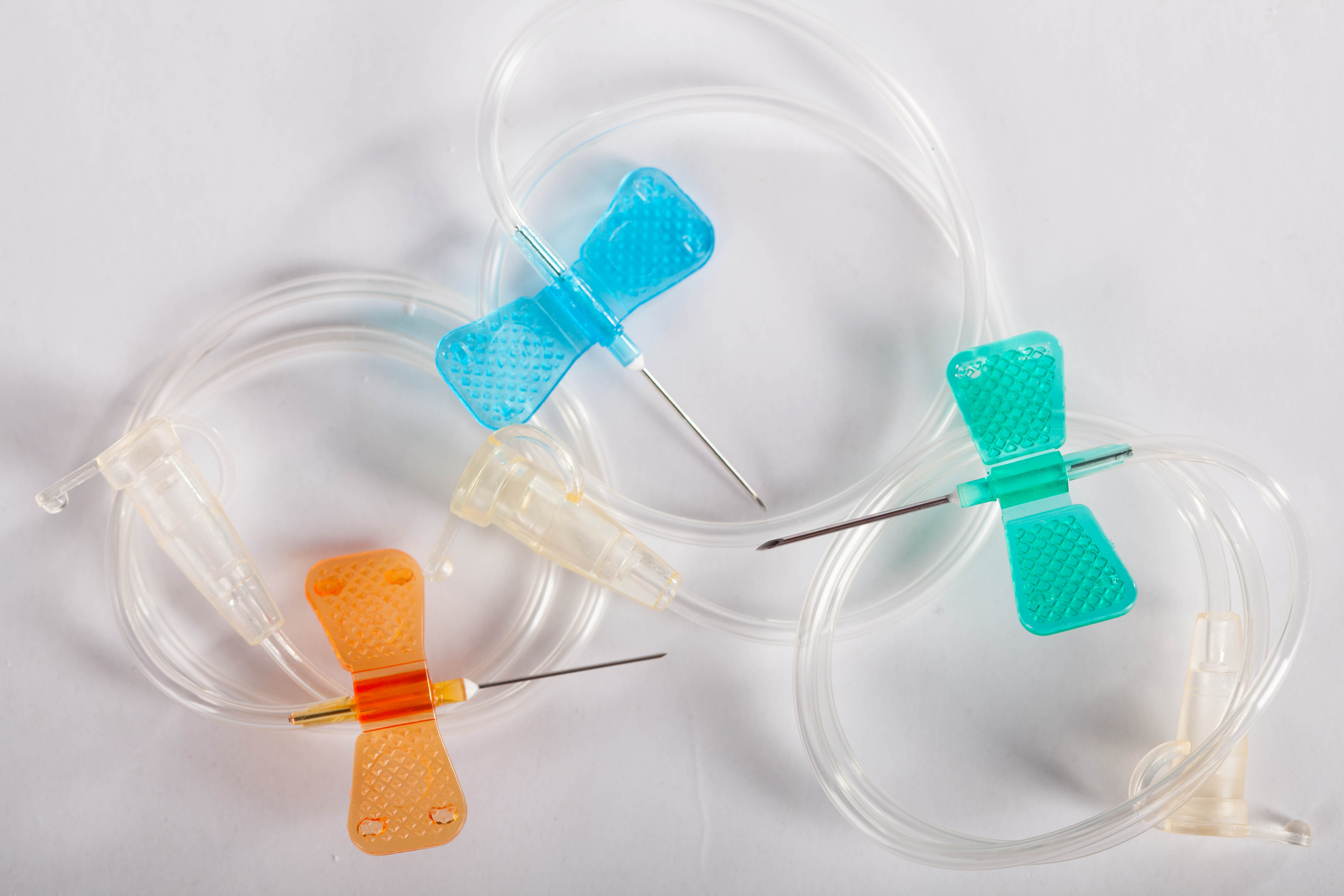
Scalp vein set trade mark IGAR
Scalp vein set trade mark IGAR – is an intravenous catheter intended for medications infusion into peripheral small veins, or for taking blood samples for analysis.
Standard color coding of sizes.
Scalp vein set is very frequently used in veterinary medicine.
Аdvantages:
- High-quality, sharp needle for minimal tissue damage.
- Due to the flexibility of the tube, the needle does not move during manipulations with the tube, which minimizes the risk of mechanical damage to the veins walls
- Flexible tube significantly reduces the risk of folding and breakage during infusion
- To ensure less pressure on the vein walls, the wings are slightly angled.
Specifications:
- The device consists of a needle, wings, flexible tube and a cannula with a protective cap.
- Wings provide effective fixation and ease of insertion into a vein.
A short needle with particularly precise surface grinding reduces trauma to the vein. - The long and transparent tube allows you to perform other procedures at a distance from the puncture site and monitor its contents.
- A protective cap provides additional convenience for multiple infusions.
- Standard color coding of sizes.
- Sterilization – ethylene oxide.
Size specification:

Size/Packaging/Morion Code:
Packaging: 1 piece in an individual blister package/100 pieces in a group package/2000 pieces in a transport box.
Application:
Scalp vein set trade mark IGAR – is designed to administer medications to peripheral small veins with intravenous infusions, or to take blood for analysis.
SVS are very often used in veterinary medicine.
- Frequent intravenous infusions of medication for from one to several days.
- Preoperative preparation and anesthesia during surgery.
- Transfusion of blood and its components.
- Conducting courses of intravenous chemotherapy for cancer patients.
- Introduction of parenteral nutrition.
- It is possible to use with any standard infusion or transfusion systems.
Shelf life:
Shelf life: 3 years
Manufacturer: “IGAR Ltd”,Ukraine
Certification:
The medical device is declared and placed on the market of Ukraine according to the requirements of Technical regulations and Conformity Assessment of medical devices, the product has a complete package of documents and packing labelling in Ukrainian language according to the national standards and Technical regulations.
Quality management system of the Manufacturer meets the requirements of DSTU EN ISO 13485.
Declaration of Conformity №9-09 / 2016-IGAR